
The transition elements, on the other hand, lose the ns electrons before they begin to lose the ( n – 1) d electrons, even though the ns electrons are added first, according to the Aufbau principle.  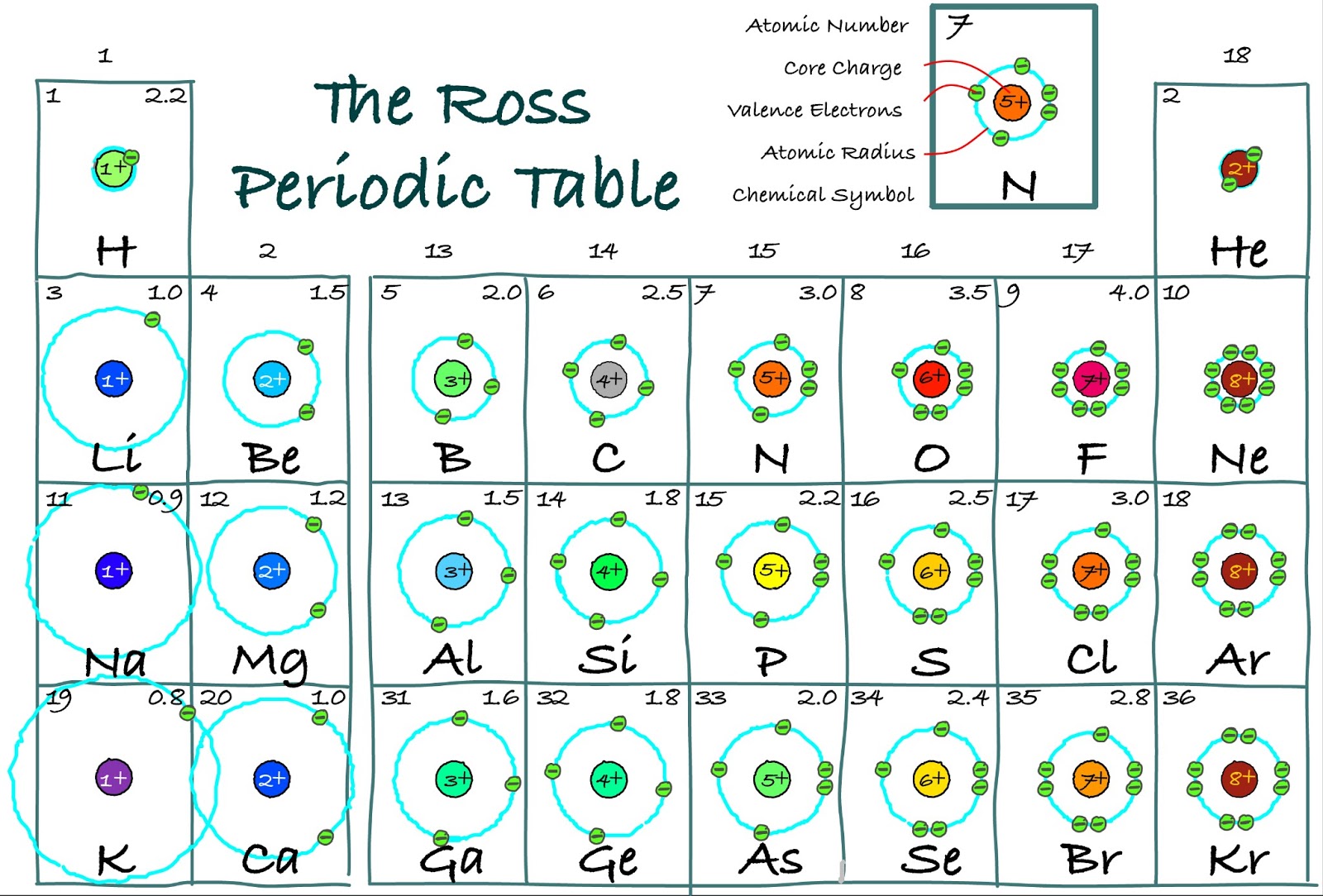
As a general rule, when the representative elements form cations, they do so by the loss of the ns or np electrons that were added last in the Aufbau process. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous -1 ions. There are seven periods in the periodic table, with each one beginning at the far left. Thus, as we would expect, the outermost or valence electrons are easiest to remove because they have the highest energies, are shielded more, and are farthest from the nucleus. Electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 16 and 17 of the Periodic Table. The locations of period 4 and period 5 transition elements are provided with green dots. Similarly, minima exist at the locations of noble or inert gases: H e, N e, A r, K r, X e, and R n. Peaks are evident at the locations of the alkali metals: L i, N a, K, R b, and C s. A black jagged line connects the radii values for elements with atomic numbers 1 through 60 on the graph. This graph is labeled Atomic Number on the horizontal axis and Radius (p m) on the vertical axis. Within each group (e.g., the alkali metals shown in purple), the trend is that atomic radius increases as Z increases. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.\): Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Number Elements Valence electrons 1 Hydrogen (H)Ģ 21 Scandium (Sc) 3 22 Titanium (Ti) 4 23 Vanadium (V) 5 24 Chromium (Cr) 6 25 Manganese (Mn) 7 26 Iron (Fe) 8 27 Cobalt (Co) 9 28 Nickel (Ni) 10 29 Copper (Cu) 11 30 Zinc (Zn) 12 31 Gallium (Ga) 3 32 Germanium (Ge) 4 33 Arsenic (As) 5 34 Selenium (Se) 6 35 Bromine (Br) 7 36 Krypton (Kr) 8 37 Rubidium (Rb) 1 38 Strontium (Sr) 2 39 Yttrium (Y) 3 40 Zirconium (Zr) 4 41 Niobium (Nb) 5 42 Molybdenum (Mo) 6 43 Technetium (Tc) 7 44 Ruthenium (Ru) 8 45 Rhodium (Rh) 9 46 Palladium (Pd) 10 47 Silver (Ag) 11 48 Cadmium (Cd) 12 49 Indium (In) 3 50 Tin (Sn) 4 51 Antimony (Sb) 5 52 Tellurium (Te) 6 53 Iodine (I) 7 54 Xenon (Xe) 8 55 Cesium (Cs) 1 56 Barium (Ba) 2 57 Lanthanum (La) 3 58 Cerium (Ce) 4 59 Praseodymium (Pr) 5 60 Neodymium (Nd) 6 61 Promethium (Pm) 7 62 Samarium (Sm) 8 63 Europium (Eu) 9 64 Gadolinium (Gd) 10 65 Terbium (Tb) 11 66 Dysprosium (Dy) 12 67 Holmium (Ho) 13 68 Erbium (Er) 14 69 Thulium (Tm) 15 70 Ytterbium (Yb) 16 71 Lutetium (Lu) 3 72 Hafnium (Hf) 4 73 Tantalum (Ta) 5 74 Tungsten (W) 6 75 Rhenium (Re) 7 76 Osmium (Os) 8 77 Iridium (Ir) 9 78 Platinum (Pt) 10 79 Gold (Au) 11 80 Mercury (Hg) 12 81 Thallium (Tl) 3 82 Lead (Pb) 4 83 Bismuth (Bi) 5 84 Polonium (Po) 6 85 Astatine (At) 7 86 Radon (Rn) 8 87 Francium (Fr) 1 88 Radium (Ra) 2 89 Actinium (Ac) 3 90 Thorium (Th) 4 91 Protactinium (Pa) 5 92 Uranium (U) 6 93 Neptunium (Np) 7 94 Plutonium (Pu) 8 95 Americium (Am) 9 96 Curium (Cm) 10 97 Berkelium (Bk) 11 98 Californium (Cf) 12 99 Einsteinium (Es) 13 100 Fermium (Fm) 14 101 Mendelevium (Md) 15 102 Nobelium (No) 16 103 Lawrencium (Lr) 3 104 Rutherfordium (Rf) 4 105 Dubnium (Db) 5 106 Seaborgium (Sg) 6 107 Bohrium (Bh) 7 108 Hassium (Hs) 8 109 Meitnerium (Mt) 9 110 Darmstadtium (Ds) 10 111 Roentgenium (Rg) 11 112 Copernicium (Cn) 12 113 Nihonium (Nh) 3 114 Flerovium (Fl) 4 115 Moscovium (Mc) 5 116 Livermorium (Lv) 6 117 Tennessine (Ts) 7 118 Oganesson (Og) 8 Free Gift for you: Interactive Periodic Table In the second period elements, the two electrons in the 1s 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. Valence Electrons Chart for All Elements Atomic Valence electrons are the electrons in the highest occupied principal energy level of an atom. (Where you will get the HD images along with the explanation). As we might predict, it becomes easier to add an electron across a series of atoms as the effective nuclear charge of the atoms increases. Values are not shown for groups 3 to 12 as well as period 7. If you want a Periodic table with Valence electrons, then visit Periodic table with Valence electrons labeled in it. The trend for electron affinity values across periods and down groups is shown in this version of the periodic table. But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons present in the outermost orbit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
